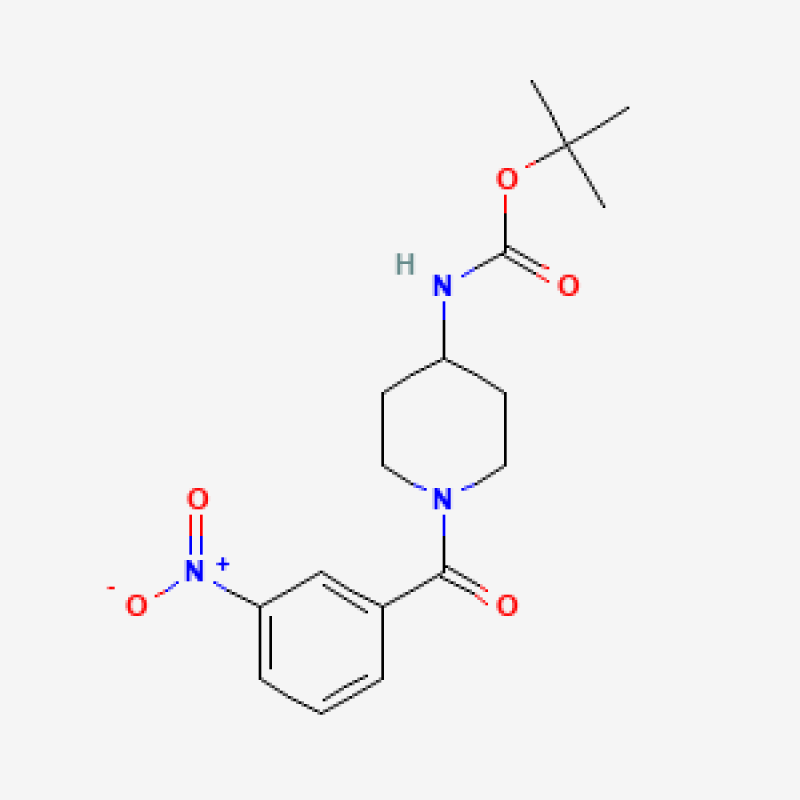
tert-Butyl 1-(3-nitrobenzoyl)piperidin-4-ylcarbamate - 1g
Short description
tert-Butyl 1-(3-nitrobenzoyl)piperidin-4-ylcarbamate
Discover the exceptional tert-Butyl 1-(3-nitrobenzoyl)piperidin-4-ylcarbamate, a versatile chemical compound with a CAS number of 1286274-65-4. Boasting a purity of 95.0% and a molecular weight of 349.3869934082031, this carbamate derivative offers a unique blend of chemical properties. Crafted with precision, it features a piperidine core and a nitrobenzoyl moiety, unlocking a world of potential applications in organic synthesis, pharmaceutical development, and beyond. Handle with care, as this compound may cause skin irritation, eye irritation, and respiratory discomfort. Ensure proper storage in a well-ventilated area and follow safety guidelines to maximize its performance in your laboratory endeavors.
- CAS: 1286274-65-4
- Purity: 95.0%
- Molecular Weight: 349.3869934082031
- Hazard Statements: H302, H315, H319, H335
- Precautionary Statements: P261, P264, P270, P271, P280, P301+P330+P331, P303+P361+P353, P304+P340, P305+P351+P338, P308+P313, P321, P362+P364, P403+P233, P405, P501
-
Procurenet Team Tshim Sha Tsui
Hong Kong 3 years
Description
tert-Butyl 1-(3-nitrobenzoyl)piperidin-4-ylcarbamate
Introducing tert-Butyl 1-(3-nitrobenzoyl)piperidin-4-ylcarbamate, a highly specialized chemical compound that holds immense potential for researchers and scientists across various fields. With its unique chemical structure and exceptional purity, this Carbamate and Impurities product, belonging to the Piperidine and Impurities subcategory, is poised to unlock new possibilities in your research endeavors.
At the heart of this compound lies a meticulously crafted molecular design, featuring a tert-butyl group, a 1-(3-nitrobenzoyl) moiety, and a piperidin-4-ylcarbamate backbone. This intricate combination of functional groups endows tert-Butyl 1-(3-nitrobenzoyl)piperidin-4-ylcarbamate with a diverse range of applications, making it a valuable asset in the realms of pharmaceutical research, chemical synthesis, and beyond.
Pharmaceutical Research
In the dynamic field of pharmaceutical research, tert-Butyl 1-(3-nitrobenzoyl)piperidin-4-ylcarbamate shines as a versatile building block. Its unique chemical structure allows for the synthesis of innovative drug candidates, targeting a wide spectrum of health conditions. Researchers can leverage the compound's properties to develop novel therapeutic agents with enhanced pharmacological profiles, improved efficacy, and reduced side effects.
The presence of the nitro group and the carbamate functionality within the molecule opens up a world of possibilities for medicinal chemists. These moieties can be strategically manipulated to modulate the compound's biological activity, enabling the exploration of new therapeutic avenues and the advancement of cutting-edge pharmaceutical solutions.
Chemical Synthesis
Beyond its pharmaceutical applications, tert-Butyl 1-(3-nitrobenzoyl)piperidin-4-ylcarbamate finds its place as a valuable reagent in the realm of chemical synthesis. Its versatile reactivity and selectivity make it a powerful tool in the hands of organic chemists, allowing for the creation of complex molecules with tailored properties.
The compound's unique structural features, such as the tert-butyl group and the nitro-substituted benzoyl moiety, can be leveraged to facilitate a wide range of synthetic transformations. These transformations can lead to the development of novel materials, intermediates, and other compounds with diverse applications in fields like materials science, agrochemicals, and beyond.
Technical Specifications
- CAS Number: 1286274-65-4
- Purity: 95.0%
- Molecular Weight: 349.3869934082031 g/mol
- InChIKey: OKHUOKJXKZFMFU-UHFFFAOYSA-N
- Hazard Statements: H302, H315, H319, H335
- Precautionary Statements: P261, P264, P270, P271, P280, P301+P330+P331, P303+P361+P353, P304+P340, P305+P351+P338, P308+P313, P321, P362+P364, P403+P233, P405, P501
Handling and Storage
tert-Butyl 1-(3-nitrobenzoyl)piperidin-4-ylcarbamate should be handled with care, as it is classified as a hazardous substance. Appropriate personal protective equipment, such as gloves, goggles, and a face mask, should be worn when working with this compound. It is essential to work in a well-ventilated area and avoid breathing in any dust, fumes, or vapors.
For optimal storage
Specifications
- Class: 14.1.5
- H statements: H302:Harmful if swallowed.H315:Causes skin irritation.H319:Causes serious eye irritation.H335:May cause respiratory irritation.
- Inchi key: InChIKey=OKHUOKJXKZFMFU-UHFFFAOYSA-N
- Molecular weight: 349.3869934082031
- Notes: This product is intended for laboratory use only, and it is not meant for human consumption.Signal word: Warning
- P statements: P261:Avoid breathing dust, fumes, gas, mist, vapours, spray. [As modified by IV ATP].P264:Wash thoroughly after handling.P270:Do not eat, drink or smoke when using this product.P271:Use only outdoors or in a well-ventilated area.P280:Wear protective glove
- Purity: 95.0%