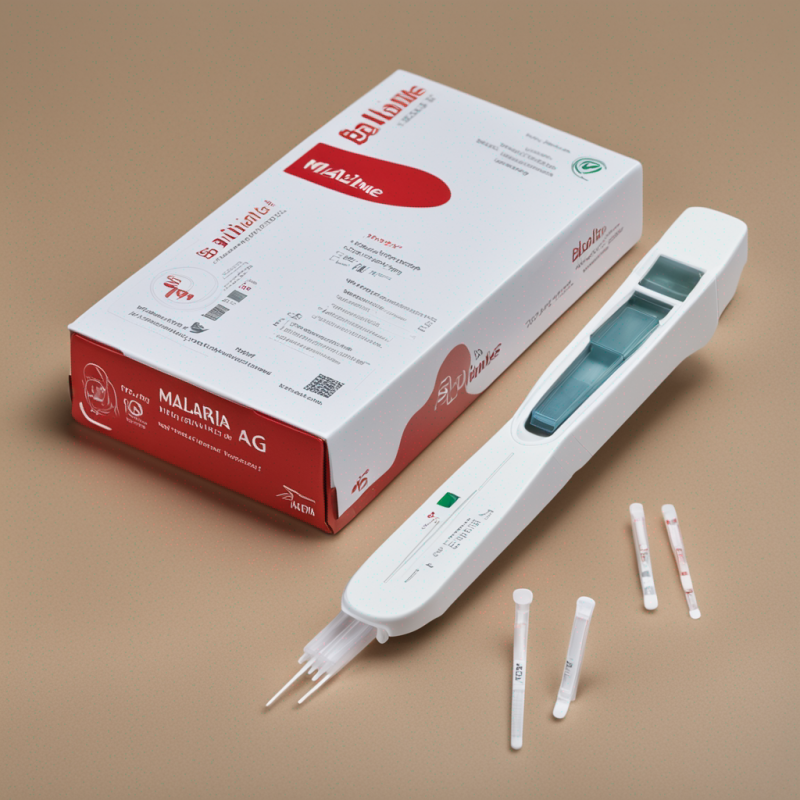
SD Bioline Malaria Ag Pf/Pan Rapid Detection Test Kit: Accurate Rapid Malaria Detection
ProcurenetShort description
The SD Bioline Malaria Ag Pf/Pan Rapid Detection Test Kit is a robust tool for quick and precise malaria detection. Implementing lateral immuno-chromatographic technology, it delivers dependable results in a timely manner. Noteworthy for its high accuracy even with lower parasitemia, it is an essential in any disease control toolkit. Every package includes 25 test devices and testing essentials, excluding disposable gloves and bio-waste disposal container. Store optimally between 1-40u2103, away from sunlight.
-
Procurenet Team Tshim Sha Tsui
Hong Kong 3 years
Description
SD Bioline Malaria Ag Pf/Pan Rapid Detection Test Kit: High-accuracy Malaria Detection
The SD Bioline Malaria Ag Pf/Pan Rapid Detection Test Kit is a cutting-edge solution developed for fast and reliable malaria detection. Engineered with the superior lateral immuno-chromatographic technology, this test kit is created to deliver remarkably accurate results in a short span of 15 minutes. The kit is optimized for substantial screenings making it highly efficient for large-scale detection.
Impressive Detection Scores
The test kit demonstrates an impressive 94.0% detection rate for low P.f. parasitemia and 91.4% for low P.v. parasitemia. With these high detection scores, the risk of false positives is greatly reduced. The zero invalidity rate of this kit lays emphasis on its reliability and trustworthiness.
All-inclusive Testing Kit
This thorough kit comprises 25 test devices, a testing buffer, specimen transfer devices, safety lancets, and alcohol swabs. Coupled with precise instructions, it ensures correct utilisation and consequently, accurate results.
Prompt and Handy
The compact and lightweight design of the kit allows for easy transportation and storage, making it optimal for use in diverse areas and critical situations. With a prompt turnaround time of just 15 minutes, immediate actions can be taken based on the results.
Long Shelf Life
The kit boasts a shelf-life of 24 months from the date of manufacture, guaranteeing its usability over an extended period and in challenging climatic conditions.
Usage Guidelines
Adherence to the manufacturer’s instructions is crucial to avoid any inaccuracies. The device should be stored away from extreme temperatures, humidity, and direct sunlight. The reagents should not be frozen. The testing area needs to be disinfected prior to the procedure and the used kits should be disposed of responsibly as per local regulations.